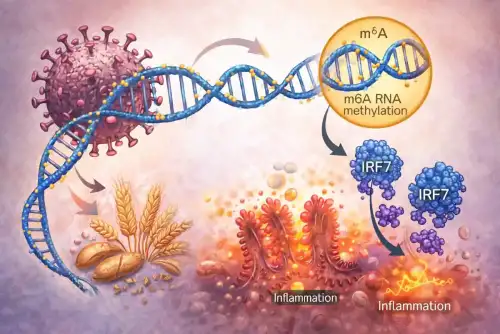
Celiac.com 03/03/2026 - Celiac disease is an immune disorder triggered by the protein gluten, which is found in wheat, barley, and rye. When someone with the condition eats gluten, their immune system mistakenly attacks the lining of the small intestine, causing inflammation and long-term health problems. While the role of gluten in activating the adaptive immune system is well known, less is understood about how the innate immune system and other cellular processes contribute to the disease.
One area of growing interest is the study of chemical changes to RNA, the molecule that carries instructions from DNA to help make proteins. One of the most common chemical changes in RNA is called N6-methyladenosine, often shortened to m6A. This modification can influence how genes are expressed in response to environmental signals, including viral infections and inflammatory triggers. The study summarized here explores how m6A RNA methylation may help regulate antiviral responses and inflammatory pathways in celiac disease.
What Is RNA Methylation?
Celiac.com Sponsor (A12):
Every cell in the body uses RNA as a messenger to convert genetic information into the proteins that carry out important functions. Sometimes, chemical tags are added to RNA molecules, and these tags can change how the RNA is read and used by the cell. One such tag is m6A, which is a small chemical group attached to specific spots on messenger RNA. The level of m6A on an RNA molecule can affect how much protein is made from that message and how the immune system responds to biological stress, including viral exposure.
How Viral Signals and Gluten Might Interact
Some viruses have been proposed as environmental triggers for autoimmune diseases, including celiac disease. In this study, researchers investigated the connection between viral signals and gluten exposure by creating experimental conditions that mimic both in laboratory-grown cells. They used a synthetic compound that acts like viral genetic material and combined it with gluten fragments to see how cells respond when “viral” stress and gluten are present together.
Importantly, they looked at a gene called Interferon Regulatory Factor 7, or IRF7, which plays a key role in antiviral immunity. Under normal circumstances, IRF7 helps the body detect and respond to viruses. The researchers found that when both the viral mimic and gluten fragments were present, the activity of IRF7 was increased more than when either component was present by itself. This suggests that the combination of viral signals and gluten could amplify certain immune pathways.
m6A Methylation Changes the Immune Signals
The study discovered that the increased activity of IRF7 in response to viral-like stress and gluten is closely tied to changes in m6A RNA methylation on the IRF7 message. In other words, the chemical tagging of the IRF7 RNA molecule appeared to help the cell produce more of the IRF7 protein when both viral signals and gluten were present. This suggests that m6A is not just a passive mark on RNA, but that it plays an active role in boosting the production of immune regulators under certain stress conditions.
This connection becomes even more interesting when the researchers looked at actual human samples. They found that people with active celiac disease had higher levels of antiviral immune activity and increased m6A RNA methylation in intestinal tissues, compared to people without the disease. This supports the idea that this mechanism may be relevant in human disease, not just in laboratory experiments.
Testing Possible Ways to Reduce the Response
Another key finding was that reducing the amount of m6A on the IRF7 RNA message led to lower levels of IRF7 protein and reduced expression of inflammatory genes downstream of IRF7. The researchers achieved this in the laboratory by silencing specific enzymes that add m6A tags, and also by using a drug known to reduce RNA methylation. When m6A levels were lowered, the heightened immune signals were dampened, suggesting that targeting RNA methylation could be a way to reduce inflammation.
Connecting Science to Human Health
This study shows that RNA methylation, and specifically m6A, is not just a background chemical process, but something that can influence how the immune system responds to environmental cues like viral stress and gluten exposure. Because people with celiac disease already have a sensitive and overactive immune response to gluten, the finding that m6A can amplify certain immune pathways may help explain why some triggers lead to more severe inflammation.
The fact that components of the immune system and the m6A machinery are elevated in people with active celiac disease suggests that this mechanism may be part of the disease process in patients, not just in cells grown in a laboratory. This opens the door to new ways of thinking about how celiac disease develops and persists, and how it might be treated in the future.
Why This Could Be Meaningful for People With Celiac Disease
For people living with celiac disease or gluten sensitivity, research like this highlights the complexity of their condition. Celiac disease is not simply a matter of eating gluten and having a reaction; it involves a web of interactions between diet, the immune system, and even how cells chemically modify their RNA. The discovery that m6A methylation can influence immune responses suggests that scientists may eventually be able to develop new strategies to calm harmful inflammation or prevent it from becoming exaggerated.
While this research does not change current treatment recommendations — the only proven therapy for celiac disease remains a strict, lifelong gluten-free diet — it does provide a deeper understanding of why the immune system behaves the way it does in this condition. In the long term, this kind of insight could lead to therapies that reduce inflammation without altering diet or that help identify why some people respond differently to environmental triggers.
In summary, the study suggests that chemical changes to RNA, such as m6A methylation, play a role in shaping the immune response to both viral signals and gluten. By affecting key regulators like IRF7, these RNA modifications may contribute to the autoimmune inflammation characteristic of celiac disease, and targeting them could one day offer new avenues for treatment.
Read more at: nature.com













Recommended Comments
There are no comments to display.
Create an account or sign in to comment
You need to be a member in order to leave a comment
Create an account
Sign up for a new account in our community. It's easy!
Register a new accountSign in
Already have an account? Sign in here.
Sign In Now