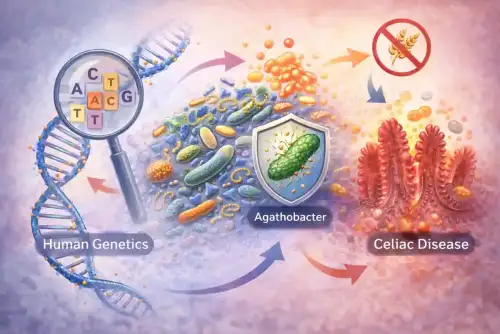
Celiac.com 04/06/2026 - Celiac disease develops when the immune system reacts abnormally to gluten, damaging the lining of the small intestine. While specific genetic markers in the human leukocyte antigen region are known to increase risk, not everyone with these genes develops the disease. Researchers have long suspected that additional environmental and biological factors influence whether celiac disease actually appears. One area of growing interest is the gut microbiome, the community of bacteria and other microorganisms that live in the digestive tract.
This large population-based study investigated how human genetic variation influences the composition and function of the gut microbiome, and whether certain microbial patterns are associated with celiac disease. By analyzing genetic data together with stool samples from thousands of participants, the researchers aimed to clarify whether specific bacteria might contribute to risk, protection, or disease progression.
Study Design and Population
Celiac.com Sponsor (A12):
The investigation was conducted using data from a large Norwegian health study that included more than twelve thousand adults of European ancestry. Participants provided stool samples for microbiome analysis and underwent genetic testing. Among them were individuals diagnosed with celiac disease, identified through medical records and screening tests.
The researchers performed a genome-wide analysis, meaning they scanned the entire genetic code of participants to identify variants associated with differences in gut bacteria. They then examined whether those same genetic patterns were linked to celiac disease. To strengthen their findings, they tested many of the associations in independent Swedish cohorts comprising over sixteen thousand additional participants.
Genetics Influences the Microbiome
The results showed that certain human genetic variants were strongly associated with the relative abundance of specific gut bacterial species. For example, genetic variation near the lactase gene influenced the presence of bacteria involved in digesting lactose. Individuals with the genetic form associated with lactose intolerance showed differences in the abundance and activity of certain Bifidobacterium species.
These findings support the idea that human genetics partly shapes the microbiome by influencing dietary processing and nutrient availability. When digestion of certain sugars is altered, it can create a different environment in the intestine, favoring the growth of some microbes over others.
A Potential Protective Bacterium in Celiac Disease
One of the most notable findings related directly to celiac disease. A specific genetic variant within the human leukocyte antigen region was associated with higher levels of a bacterial species called Agathobacter sp000434275. Interestingly, individuals carrying this genetic variant had a lower risk of developing celiac disease.
Further analysis revealed that participants with higher levels of this bacterial species were less likely to have celiac disease. This association was observed in cross-sectional data, meaning it reflected patterns seen at a single point in time rather than over many years. Statistical modeling suggested that celiac disease itself might reduce the abundance of this bacterium, although it remained unclear whether low levels of the bacterium contribute to disease development or result from the disease process.
In simple terms, people with more of this particular microbe in their gut tended to have a lower prevalence of celiac disease. However, the study could not definitively prove whether the bacterium actively protects against disease or whether the inflammatory environment of celiac disease reduces its presence.
Microbial Function Matters, Not Just Species
Beyond identifying individual bacterial species, the researchers examined functional pathways within the microbiome. These pathways describe the biochemical activities performed by gut bacteria, such as nutrient metabolism or energy production.
Certain genetic variants were associated with differences in microbial functional modules. For example, individuals with lactose intolerance–associated genetic variants showed increased activity in pathways related to phosphate transport and energy metabolism. This suggests that genetic differences can influence not only which bacteria are present, but also how actively they carry out specific metabolic tasks.
These functional differences may have broader implications for immune signaling, nutrient absorption, and inflammation within the intestine. Since celiac disease involves an abnormal immune response in the gut, shifts in microbial metabolic activity could potentially influence how the immune system behaves.
Replication Strengthens the Findings
To ensure the reliability of their results, the investigators tested many of their genetic–microbiome associations in independent populations from Sweden and Finland. Most of the key associations were successfully replicated, meaning similar patterns were observed in separate groups of participants.
Replication is critical in large genetic studies because it reduces the likelihood that findings are due to chance. The consistency across multiple populations strengthens confidence that the identified relationships between genes and microbes are biologically meaningful.
Interpreting Cause and Effect
Although the study identified strong associations between genetic variants, microbial composition, and celiac disease risk, it remains difficult to determine direct causation. The researchers used statistical approaches designed to estimate whether changes in microbiome composition might influence disease risk, or whether the disease itself alters the microbiome.
For the bacterial species linked to reduced celiac disease risk, there was some evidence suggesting that having celiac disease could lower its abundance. However, the possibility that the bacterium might also play a protective role could not be ruled out. Longitudinal studies following individuals over time will be needed to clarify this relationship.
Why This Study Matters for People with Celiac Disease
This research highlights the complex interaction between human genetics and the gut microbiome in celiac disease. While gluten exposure remains the essential trigger, the findings suggest that certain microbial environments may influence whether genetically susceptible individuals develop the disease.
If specific bacteria such as Agathobacter species are eventually shown to play a protective role, future therapies might aim to support their growth through diet, probiotics, or other microbiome-targeted approaches. In addition, understanding how genetics shapes the microbiome could help explain why individuals respond differently to gluten exposure or experience varying degrees of inflammation.
At present, strict adherence to a gluten-free diet remains the only proven treatment for celiac disease. However, this study opens the possibility that modifying the gut microbiome could someday complement dietary therapy. By identifying bacterial species and metabolic pathways linked to disease risk, researchers are moving closer to understanding the broader biological context in which celiac disease develops.
Conclusion
This large genetic and microbiome analysis provides valuable insight into how human DNA influences gut bacteria and how these microbes may be connected to celiac disease. While more research is needed to determine causation, the findings suggest that the microbiome is not merely a bystander but may interact closely with genetic susceptibility. For individuals with celiac disease, this work offers hope that future prevention or treatment strategies might extend beyond gluten avoidance and target the intestinal ecosystem itself.
Read more at: nature.com








Recommended Comments
There are no comments to display.
Create an account or sign in to comment
You need to be a member in order to leave a comment
Create an account
Sign up for a new account in our community. It's easy!
Register a new accountSign in
Already have an account? Sign in here.
Sign In Now